Main
Bipolar disorder is a chronic, relapsing–remitting illness characterized by episodes of mania, hypomania and depression and is associated with substantial functional impairment and increased mortality. According to the major clinical guidelines, lithium is widely regarded as the ‘gold standard’ mood stabilizer for long-term management1,2,3. Indeed, a large body of research has shown the effectiveness of lithium treatment in reducing psychiatric rehospitalization risk, mood-episode recurrence and suicide risk4,5,6,7. However, many patients are unable to sustain long-term lithium treatment due to either inadequate therapeutic response or intolerance to adverse effects8,9, creating a major clinical dilemma for which high-quality evidence remains limited. In addition, lithium prescription rates have fallen substantially in recent years10.
Approximately one in five patients experiences relapse despite adequate lithium serum concentrations and might therefore require augmentation with other pharmacotherapies9. Although recent network meta-analyses support the efficacy of combining antipsychotics with lithium or valproate for acute mania11,12, their ability to prevent long-term relapse remains unclear. Existing randomized controlled trials (RCTs) vary substantially in methodology, include few combination regimens and rely on short follow-up periods11,12. Consequently, guidelines provide limited recommendations on specific augmentation strategies when lithium alone is insufficient, and real-world prescribing often relies on clinical experience rather than evidence-based comparisons1,2,3,13.
Lithium cessation, for example, due to renal or thyroid dysfunction, represents a distinct and equally important clinical challenge. Up to half of individuals discontinue lithium because of adverse effects8. For these patients, clinicians require alternative long-term pharmacotherapeutic approaches that mimic the benefits of lithium. Yet evidence on treatment selection after lithium discontinuation is remarkably sparse. Previous observational studies have examined broad medication classes14,15,16,17,18 but have not evaluated the comparative effectiveness of specific combinations of antipsychotics and mood stabilizers in individuals who have discontinued lithium use. As a result, this vulnerable subgroup, although common in clinical practice, remains largely understudied.
Long-acting injectable (LAI) antipsychotics also merit closer examination. RCTs suggest that LAIs may improve maintenance treatment in bipolar disorder, particularly in patients with poor adherence19,20,21, but head-to-head comparisons with lithium are lacking. Previous cohort studies indicated potential benefits in bipolar disorder18, yet sample sizes have been too small to reliably evaluate individual LAIs or their combinations with mood stabilizers.
Altogether, these limitations highlight the need for large-scale, longitudinal, population-based data to identify which pharmacotherapeutic strategies are most effective when lithium is ineffective or discontinued. Register-based nationwide cohorts offer a unique opportunity to assess comparative effectiveness in unselected populations while capturing long-term treatment trajectories and relapse outcomes22. Given the high prevalence of polypharmacy in bipolar disorder23 and the large number of clinically used medication pairings, adequately powered analyses require sample sizes far exceeding those available in existing RCTs or single-country observational studies.
In this study, we addressed these evidence gaps by leveraging two extensive, nationwide cohorts from Sweden and Finland. Our aims were (1) to evaluate the comparative effectiveness of all commonly used pharmacotherapies (that is, monotherapies and combination regimens) on the risk of psychiatric hospitalization; (2) to identify pharmacotherapies that have a lower hospitalization risk than lithium monotherapy does, offering potential options when lithium monotherapy is insufficient; and (3) to assess treatment strategies among patients who discontinue lithium. By using within-individual models that eliminate selection bias22 and pooling results via meta-analysis, we provide a comprehensive evaluation of long-term pharmacotherapeutic strategies for bipolar disorder when lithium is inadequate or discontinued.
Results
Sociodemographic and clinical characteristics
We identified 105,495 patients with bipolar disorder (65,607 women, mean (s.d.) age: 44.23 (18.79) years, lithium users: 27,058, mean (s.d.) follow-up: 9.09 (5.15) years) from Sweden and 60,045 individuals (33,859 women; mean (s.d.) age: 41.7 (15.8) years, lithium users: 7,700, mean (s.d.) follow-up: 9.45 (6.01) years) from Finland (Table 1). Over the total available follow-up, 36,732 patients had psychiatric hospitalizations in the Swedish cohort (119,059 psychiatric hospitalizations in total) and 26,464 in the Finnish cohort (93,922 in total). Cumulative incidence of psychiatric hospitalizations over the follow-up across the two cohorts is depicted in Supplementary Fig. 1. Compared with those in the Swedish cohort, patients in the Finnish cohort were younger, fewer were women, and they had greater rates of recent sickness absence and disability pensions. Newly diagnosed (incident) bipolar disorder was slightly less common in Finland, and Finnish patients had a lower prevalence of anxiety disorders but a higher prevalence of bipolar mania and comorbid substance use disorders.
Comparative effectiveness of all pharmacotherapeutic strategies
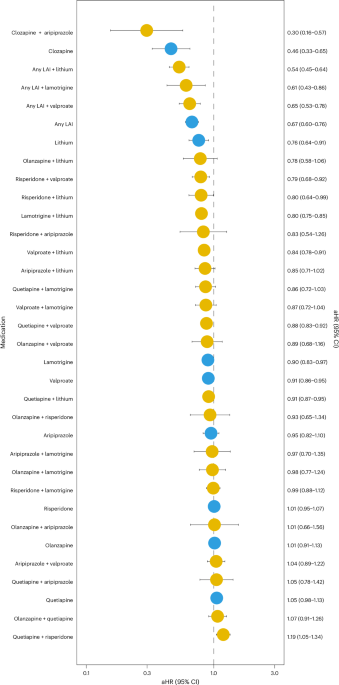
In a within-individual meta-analysis pooling data from Sweden and Finland and comparing each treatment period with the patient’s own medication-free periods (Fig. 1), clozapine plus aripiprazole was associated with the lowest risk of psychiatric hospitalization (meta-analysis adjusted hazard ratio (aHR) = 0.30, 95% confidence interval (CI) = 0.16–0.57, false discovery rate (FDR)-corrected P = 0.0009), although this estimate was based on only 3 events in Sweden and 11 in Finland (total number of users = 221). Additional regimens associated with the lowest psychiatric hospitalization risk included clozapine monotherapy (aHR 0.46, 95% CI 0.33–0.65, FDR-corrected P < 0.0001), LAI antipsychotic plus lithium (aHR 0.54, 95% CI 0.45–0.64, FDR-corrected P < 0.0001), LAI antipsychotic plus lamotrigine (aHR 0.61, 95% CI 0.43–0.86, FDR-corrected P = 0.012), LAI antipsychotic plus valproate (aHR 0.65, 95% CI 0.53–0.78, FDR-corrected P < 0.0001), LAI antipsychotic monotherapy (aHR 0.67, 95% CI 0.60–0.76, FDR-corrected P < 0.0001) and lithium monotherapy (aHR 0.76, 95% CI 0.64–0.91, FDR-corrected P = 0.0078). The results from individual cohorts are shown in Supplementary Tables 1 and 2. Across the two cohorts, the rank ordering of treatment-specific aHR estimates was highly aligned (Spearman’s rho = 0.79).
Monotherapies in blue; combinations in yellow. The results are drawn from the random-effects meta-analysis of the two cohorts (Swedish cohort, N = 105,495; Finnish cohort, N = 60,045).
We conducted several sensitivity analyses to assess the robustness of these findings. Omitting the first 30 days of each exposure produced nearly identical estimates with the abovementioned findings (Supplementary Fig. 2). In addition, the abovementioned findings from within-individual analyses largely aligned with the results from traditional between-individual analyses (Pearson’s r = 0.78; Supplementary Figs. 3 and 4). The only major difference between these analyses was that LAI plus lithium did not confer a statistically significant risk reduction (aHR 1.05, 95% CI 0.77–1.44). In a sensitivity analysis (Supplementary Fig. 5) excluding individuals who later converted to schizophrenia (International Classification of Diseases, Tenth Revision (ICD-10): F20) or schizoaffective disorder (ICD-10: F25), LAI antipsychotic plus lithium demonstrated the lowest psychiatric hospitalization risk (aHR 0.56, 95% CI 0.45–0.70), followed by clozapine monotherapy (aHR 0.66, 95% CI 0.51–0.85), with lithium monotherapy at an aHR of 0.76 (95% CI 0.62–0.92). When focusing on incident bipolar disorder cases (48,893 in Sweden; 26,395 in Finland), the results were consistent with those of the overall cohorts (Supplementary Fig. 6): LAI antipsychotic plus lithium yielded the lowest psychiatric hospitalization risk (aHR 0.45, 95% CI 0.31–0.65), whereas lithium monotherapy had an aHR of 0.82 (95% CI 0.66–1.02). The results for incident bipolar disorder in each cohort are presented in Supplementary Tables 3 and 4. For all-cause hospitalizations (Supplementary Fig. 7), the combination of LAI antipsychotic plus lithium conferred the lowest risk (aHR 0.64, 95% CI 0.56–0.74), followed by clozapine monotherapy (aHR 0.74, 95% CI 0.60–0.91). In this analysis, the association between lithium monotherapy and the aHR was 0.79 (95% CI 0.72–0.87). Finally, among individual LAIs and their combinations (Supplementary Fig. 8), the lowest risk of psychiatric hospitalization was observed when the first-generation antipsychotic LAI was combined with lithium (aHR 0.41, 95% CI 0.31–0.54). Among the lithium plus LAI regimens, the aripiprazole–lithium combination was associated with the least favorable outcomes (aHR 1.26, 95% CI 0.68–2.31).
Effectiveness of pharmacotherapeutic strategies compared with lithium monotherapy
Compared with lithium monotherapy (Fig. 2), only the combination of clozapine plus aripiprazole (aHR 0.42, 95% CI 0.22–0.80, FDR-corrected P = 0.016), clozapine monotherapy (aHR 0.61, 95% CI 0.49–0.75, FDR-corrected P < 0.0001) and LAI antipsychotics plus lithium (aHR 0.70, 95% CI 0.59–0.84, FDR-corrected P = 0.0003) were related to a significantly lower risk of psychiatric hospitalization. Note, however, that the periods of combination of clozapine plus aripiprazole incorporated only 3 events in the Swedish cohort and 11 in the Finnish cohort. The results for individual cohorts are shown in Supplementary Tables 5 and 6.
Monotherapies in blue; combination treatments in yellow; nonuse of antipsychotics or mood stabilizers in black. The results are drawn from the random-effects meta-analysis of the two cohorts (Swedish cohort, N = 105,495; Finnish cohort, N = 60,045).
Comparative effectiveness of pharmacotherapeutic strategies in lithium discontinuation
Among 15,568 Swedish and 5,077 Finnish patients who discontinued lithium (Fig. 3), the combination of an LAI antipsychotic and valproate yielded the lowest point estimate for the risk of psychiatric hospitalization (aHR 0.41, 95% CI 0.19–0.92; uncorrected P = 0.031), but this finding was supported by only 3 events during the exposure periods in the Swedish cohort and 16 in the Finnish cohort (Supplementary Tables 7 and 8) and did not survive FDR correction. By contrast, a few other regimens, namely, quetiapine plus lamotrigine (aHR 0.64, 95% CI 0.51–0.79; FDR-corrected P = 0.001), olanzapine plus valproate (aHR 0.65, 95% CI 0.50–0.84; FDR-corrected P = 0.014) and risperidone plus valproate (aHR 0.63, 95% CI 0.42–0.93; uncorrected P = 0.020; FDR-corrected P = 0.15), were associated with a lower hospitalization risk, and in the case of quetiapine plus lamotrigine and olanzapine plus valproate, they remained statistically significant after FDR adjustment. The association between clozapine use and the risk of psychiatric hospitalization was not statistically significant (aHR 0.60, 95% CI 0.30−1.22; FDR-corrected P = 0.43). Notably, there were only 35 clozapine users in the Swedish cohort and 15 users in the Finnish cohort (Supplementary Tables 7 and 8).
Monotherapies in blue; combination treatments in yellow. The results are drawn from the random-effects meta-analysis of the two cohorts (Swedish cohort, N = 15,568; Finnish cohort, N = 5,077).
Discussion
In this extensive register-based study of more than 160,000 patients drawn from Swedish and Finnish nationwide cohorts, we evaluated long-term pharmacotherapy strategies for bipolar disorder via within-individual analyses and random-effects meta-analyses. Across the two cohorts, clozapine-centered regimens conferred the greatest reduction in psychiatric rehospitalization risk, followed by the addition of LAI antipsychotics to lithium treatment. Compared with nonuse of antipsychotics or mood stabilizers, monotherapy with LAI antipsychotics was associated with a 33% reduction in psychiatric hospitalization risk. After the abovementioned regimens, lithium monotherapy ranked among the most effective treatments, outperforming other mood stabilizer monotherapies. These findings largely remained across multiple sensitivity analyses, including models using all-cause hospitalizations as the outcome, omission of the first 30 days of each exposure, restriction to incident cases, and exclusion of individuals who later converted to schizophrenia. Although treatment changes may reflect factors unrelated to lithium response, the within-individual design minimizes confounding by comparing medication periods within the same patient. Any remaining time-varying confounding, such as clinical deterioration leading to treatment escalation, would tend to bias results against intensified or combination regimens, rendering our estimates conservative.
Our results highlight the importance of conducting RCTs on clozapine to assess its efficacy in bipolar disorder, given the scant evidence on its use currently captured in guidelines1,13,24. Among lithium discontinuers, the small number of clozapine users limited statistical power, resulting in a nonstatistically significant association between clozapine use and psychiatric hospitalization risk. Therefore, whether a clozapine-centered regimen constitutes an effective alternative for lithium discontinuation remains elusive, despite our findings supporting its use when lithium monotherapy is inadequate. Nevertheless, the superiority of clozapine over lithium may also partially stem from regular contact in healthcare via blood tests.
Although major consensus statements recommend clozapine only as a third- or fourth-line treatment for refractory bipolar disorder1,13,24, largely owing to limited randomized data and its off-label status in this context25, clinical experience and small-scale studies suggest broader potential. Specifically, several uncontrolled trials and case series have shown that clozapine can ameliorate acute manic and mixed symptoms in patients unresponsive to previous therapies, including lithium13,26,27,28,29,30. Furthermore, although evidence for clozapine maintenance treatment remains sparse, an open-label study of 38 treatment-resistant patients with bipolar disorder or schizoaffective disorder demonstrated that adjunctive clozapine produced superior mood stabilization over 1 year compared with treatment as usual31.
Notably, clozapine combined with aripiprazole emerged as the most effective regimen in our analysis compared with lithium monotherapy. Interestingly, the association of clozapine plus aripiprazole with the lowest rehospitalization risk parallels case series documenting substantial improvement in psychotic mania resistant to other second-generation antipsychotics32. Although replication is needed due to the small number and events in our dataset, interestingly, the same pharmacotherapeutic combination also represents the most effective antipsychotic pairing in treatment-refractory schizophrenia33.
Although findings from both observational studies and RCTs have provided evidence supporting the use of LAIs in bipolar disorder7,13,18, our findings further elucidate their potential role in patients with inadequate response to or intolerance of lithium, as these aspects have been unclear in the literature. In particular, adjunctive risperidone LAI has been shown to outperform placebo in patients maintained on lithium or valproate34 and, in a separate placebo-controlled RCT, to confer superior relapse prevention when used as standalone maintenance therapy35. Our within-individual analyses both corroborate and extend these findings: the addition of any LAI to lithium periods was associated with a 30% lower risk of psychiatric hospitalization than the same patients’ lithium monotherapy intervals. However, the same benefit was not observed with the add-on use of oral formulations of second-generation antipsychotics together with lithium. Taken together, these findings highlight the need to address medication adherence in the management of bipolar disorder.
Among lithium discontinuers, the combination of any LAI antipsychotic with valproate yielded the lowest rehospitalization point estimate. Note, however, that this finding did not survive FDR adjustment, underscoring the need for further studies to validate optimal LAI–mood stabilizer pairings in this population. Indeed, the limited number of events in our lithium discontinuation cohort precluded the demonstration of a statistically significant reduction in hospitalization risk for LAI antipsychotics overall, which contrasts with previous observational reports18.
Among lithium discontinuers, combinations of quetiapine plus lamotrigine and olanzapine plus valproate were strongly associated with reduced psychiatric hospitalization, findings not previously supported by observational studies or RCTs. Clinical guidelines offer only broad recommendations for antipsychotic augmentation and valproate use without specifying particular pairings for this population1,13,24. In our register-based cohort, quetiapine plus lamotrigine and olanzapine plus valproate each conferred significantly lower rehospitalization rates than did periods off both medication classes, whereas none of these agents alone achieved a statistically significant effect, suggesting synergistic benefits. Patients who discontinue lithium may represent a more treatment-resistant or comorbid subgroup. By definition, their treatment has failed, or they cannot tolerate the gold-standard mood stabilizer, and their unique pathophysiology may render them particularly responsive to specific drug combinations.
The strengths of this study include the use of unselected national cohorts, follow-up of up to 23 years and a within-individual design that controls for time-invariant confounders and eliminates selection bias. However, residual confounding by protopathic bias cannot be ruled out. Nevertheless, since pharmacotherapy is often initiated or intensified at times of symptom exacerbation, indication severity (rather than the indication per se) can introduce confounding factors that make combination regimens appear less effective than they are. Thus, the observed associations between specific combination regimens and lower hospitalization risk compared with lithium monotherapy should be interpreted cautiously as residual bias related to treatment escalation during symptom exacerbation cannot be excluded. Furthermore, psychiatric hospitalization, our primary outcome, serves as a proxy for severe relapse rather than a direct measure of symptom recurrence or functional status. Therefore, studies using more granular outcomes in the future are needed. In addition, hospital admissions for any psychiatric diagnosis do not distinguish symptom types, and we lack data to confirm treatment resistance among medication users. Register-based data lack detailed clinical information, such as symptom severity, adherence and reasons for treatment changes. In addition, our findings pertain to long-term outcomes rather than acute treatment effects and do not account for social support or nonpharmacologic interventions. Finally, although healthcare delivery between Sweden and Finland is relatively similar, the generalizability of the findings to markedly different healthcare systems may be limited.
Conclusions
We found that clozapine-based regimens and lithium accompanied by LAI antipsychotics demonstrated a lower risk of psychiatric hospitalization compared with lithium monotherapy. Among patients who discontinued lithium, only specific antipsychotic–mood stabilizer combinations were related to reduced psychiatric hospitalization risk. Prospective trials are needed to decipher the efficacy of these regimens.
Method
Study design and data acquisition
The study drew on two nationwide, register-based cohorts from Sweden and Finland, each constructed using harmonized inclusion and exclusion criteria. The formations of both cohorts are described elsewhere6,7.
None of the Swedish or Finnish cohorts used in this study is publicly accessible due to national data protection legislation governing sensitive health information. Ethical approval was obtained from the Regional Ethics Board of Stockholm and the Swedish Ethical Review Authority (2007/762-31; 2024-08708-02) and from the Finnish National Institute for Health and Welfare, the Social Insurance Institution of Finland, the Finnish Centre for Pensions, and Statistics Finland (permissions THL/5279/14.06.00/2023; 31/522/2019; 19023; TK-53-569-19). These permits authorized the use of pseudonymized register data for this study under secure processing environments. Because all the data were drawn from administrative registers without direct participant contact, informed consent was waived under both countries’ regulations.
Both cohorts comprised all individuals aged up to 65 years at the first recorded treatment contact for bipolar disorder (ICD-10: F30–F31) in inpatient or outpatient settings. Data regarding ethnicity were not available in the registers of the two cohorts. The Swedish sample included all such patients documented in the National Patient Register (inpatient and specialized outpatient care) or the MiDAS Register (disability pensions and sickness absences) between 1 July 2006 and 31 December 2021. The Finnish cohort similarly included patients identified via the Hospital Discharge Register of the National Institute for Health and Welfare, alongside sickness absence and disability pension records from the Social Insurance Institution and the Finnish Centre for Pensions, between 1 January 1996 and 31 December 2018. The data contained no missingness for exposures or outcomes. Loss of follow-up was either the end of available follow-up in the registers (31 December 2021), emigration, death or conversion to schizophrenia (ICD-10: F20–F29), whichever occurred first.
Exposure
Treatment exposures included mood stabilizers (Anatomical Therapeutic Chemical (ATC) codes N03AF01 (valproate), N03AG01 (lamotrigine), N03AX09 (lithium), N05AN01 (carbamazepine)), antipsychotics (ATC N05A, excluding lithium) and their various combinations. Antipsychotics were separately modeled as oral versus LAIs, but owing to the low statistical power of specific drugs, LAIs were pooled. For each comparison, the reference period was defined as nonuse of both mood stabilizers and antipsychotics (for example, valproate use was contrasted with nonuse of either class) or comparison with lithium monotherapy. Medication use periods were reconstructed from national prescription purchase data via the PRE2DUP method, which models sliding averages of defined daily doses, quantities dispensed, individual refill patterns, hospital inpatient days and potential stockpiling36.
Outcomes and statistical analyses
The primary outcome was all-cause psychiatric hospitalization (any ICD-10 F diagnosis). We conducted three successive Cox models that are detailed in the subsequent paragraphs. Descriptions of within-individual and traditional between-individual analyses are provided in Supplementary Fig. 9. In the within-individual models, each patient acts as his or her own control to eliminate time-invariant confounding factors37. The outcomes in the models were modeled as recurring occurrences. The follow-up time was reset to zero after each outcome (that is, psychiatric hospitalization), and the models were adjusted for the sequence of treatments, time since cohort entry and time-varying use of other psychotropics (antidepressants, benzodiazepines and related agents, attention deficit hyperactivity disorder medications, and medications for substance use disorders). Within-individual analyses were conducted in SAS version 9.4, and the resulting aHRs with 95% CIs were combined in R version 4.4.2 via the meta and metafor packages. Meta-analyses were conducted via random-effects models with restricted maximum likelihood estimation and Wald-type confidence intervals to account for between-study heterogeneity. Finally, Benjamini–Hochberg FDR correction was applied to control for multiple comparisons of the two-tailed P values38.
In the first within-individual analysis, each monotherapy and combination regimen was compared with periods of nonuse of mood stabilizers and antipsychotics to identify strategies associated with the lowest hospitalization risk in the entire cohort and to benchmark the performance of lithium. To test the robustness of these findings, we conducted multiple sensitivity analyses, and consistency across these sensitivity analyses was interpreted as evidence of robust, generalizable treatment effects. We first re-ran the within-individual models after excluding the first 30 days of each exposure (and nonexposure) period22, thereby reducing the risk of protopathic bias resulting from treatment initiation during acute deterioration39 as well as potential carry-over effects of previous medication use to subsequent nonuse periods. Next, we performed traditional between-individual Cox regression modeling, adjusted for age, sex, incident bipolar (yes versus no), type of first bipolar diagnosis (mania, hypomania, depression, mixed versus unknown/other), duration of inpatient stay at first diagnosis (versus no stay, outpatients), previous use of LAIs, previous use of lithium, diagnosis of substance use disorders (ICD-10: F10-F19), cardiovascular diseases (I00–I99), personality disorders (F60–F63), previous suicide attempts (X60–X84, Y10–Y34), disability pension (yes versus no), and use of antidepressants (ATC N06A), benzodiazepines and related drugs (N05BA, N05CD, N05CF) and attention deficit hyperactivity disorder drugs (N06BA), to assess whether our findings are driven solely by between-patient differences in case mix. Third, we excluded individuals who subsequently converted to schizophrenia (ICD-10: F20) or schizoaffective disorder (F25) to determine whether diagnostic shifts influenced our results. We then restricted the analysis to incident cases, patients at first bipolar diagnosis without antipsychotic or mood stabilizer exposure in the preceding year, to guard against survival bias. Next, we repeated the primary within-individual models using all-cause hospitalization as the outcome to examine whether the results extend beyond psychiatric admissions. Finally, differences in the comparative effectiveness of individual LAI antipsychotics and their combinations were evaluated to identify the most effective regimen.
A second within-individual analysis compared each alternative pharmacotherapy with patients’ own periods of lithium monotherapy. The purpose of this analysis was to identify pharmacotherapeutic strategies that could offer superior effectiveness when lithium alone is insufficient.
In the third within-individual analysis, comparative effectiveness was assessed among patients who had maintained lithium therapy for ≥6 months and then discontinued it for at least 30 days. During these lithium-free intervals, exposure to antipsychotics, mood stabilizers and their combinations was contrasted with periods of nonuse of mood stabilizers and antipsychotics, with follow-up censored at death, at the end of the study or upon reinitiation of lithium.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data are not publicly available. The Swedish data used in this study cannot be made publicly available due to privacy regulations. According to the General Data Protection Regulation, the Swedish law SFS 2018:218, the Swedish Data Protection Act, the Swedish Ethical Review Act and the Public Access to Information and Secrecy Act, these types of sensitive data can only be made available for specific purposes, including research, that meets the criteria for access to this type of sensitive and confidential data as determined by a legal review. Readers may contact E.M.-R. (ellenor.mittendorfer-rutz@ki.se) regarding the data. The Finnish data analyzed in this study are not publicly available due to participant privacy and security concerns. Access to the data can be obtained from the Finnish Social and Health Data Permit Authority (FinData).
Code availability
No custom computer code was developed for the analyses in this study. All statistical analyses were performed using standard SAS 9.4 procedures and established R packages (meta, metafor). An R version of the PRE2DUP method is available at https://github.com/piavat/PRE2DUP-R.
References
-
Yatham, L. N. et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 20, 97–170 (2018).
-
American Psychiatric Association. Practice guideline for the treatment of patients with bipolar disorder (revision). Am. J. Psychiatry 159, 1–50 (2002).
-
Kendall, T., Morriss, R., Mayo-Wilson, E. & Marcus, E. Assessment and management of bipolar disorder: summary of updated NICE guidance. BMJ 349, g5673 (2014).
-
Fountoulakis, K. N., Tohen, M. & Zarate, C. A. Lithium treatment of bipolar disorder in adults: a systematic review of randomized trials and meta-analyses. Eur. Neuropsychopharmacol. 54, 100–115 (2022).
-
Cipriani, A., Hawton, K., Stockton, S. & Geddes, J. R. Lithium in the prevention of suicide in mood disorders: updated systematic review and meta-analysis. BMJ 346, f3646 (2013).
-
Ermis, C. et al. Real-world effectiveness of pharmacological maintenance treatment of bipolar depression: a within-subject analysis in a Swedish nationwide cohort. Lancet Psychiatry 12, 198–207 (2025).
-
Lähteenvuo, M., Paljärvi, T., Tanskanen, A., Taipale, H. & Tiihonen, J. Real-world effectiveness of pharmacological treatments for bipolar disorder: register-based national cohort study. Br. J. Psychiatry 223, 456–464 (2023).
-
Öhlund, L. et al. Reasons for lithium discontinuation in men and women with bipolar disorder: a retrospective cohort study. BMC Psychiatry 18, 37 (2018).
-
Peselow, E. D., Fieve, R. R., Difiglia, C. & Sanfilipo, M. P. Lithium prophylaxis of bipolar illness: the value of combination treatment. Br. J. Psychiatry 164, 208–214 (1994).
-
Poranen, J. et al. Twenty-year medication use trends in first-episode bipolar disorder. Acta Psychiatr. Scand. 146, 583–593 (2022).
-
Huang, W., He, S., Liu, M. & Xu, J. Comparative efficacy, safety, and tolerability of pharmacotherapies for acute mania in adults: a systematic review and network meta-analysis of randomized controlled trials. Mol. Psychiatry 30, 838–847 (2025).
-
Kishi, T. et al. Mood stabilizers and/or antipsychotics for bipolar disorder in the maintenance phase: a systematic review and network meta-analysis of randomized controlled trials. Mol. Psychiatry 26, 4146–4157 (2021).
-
Fountoulakis, K. N. et al. The CINP guidelines on the definition and evidence-based interventions for treatment-resistant bipolar disorder. Int. J. Neuropsychopharmacol. 23, 230–256 (2020).
-
Wingård, L. et al. Monotherapy vs. combination therapy for post mania maintenance treatment: a population based cohort study. Eur. Neuropsychopharmacol. 29, 691–700 (2019).
-
Altamura, A. C. et al. Quetiapine and classical mood stabilizers in the long-term treatment of bipolar disorder: a 4-year follow-up naturalistic study. J. Affect. Disord. 110, 135–141 (2008).
-
Hochman, E., Krivoy, A., Schaffer, A., Weizman, A. & Valevski, A. Antipsychotic adjunctive therapy to mood stabilizers and 1-year rehospitalization rates in bipolar disorder: a cohort study. Bipolar Disord. 18, 684–691 (2016).
-
Patel, N. C., Crismon, M. L. & Pondrom, M. Rehospitalization rates of patients with bipolar disorder discharged on a mood stabilizer versus a mood stabilizer plus an atypical or typical antipsychotic. J. Behav. Health Serv. Res. 32, 438–445 (2005).
-
Holm, M., Tanskanen, A., Lähteenvuo, M., Tiihonen, J. & Taipale, H. Comparative effectiveness of mood stabilizers and antipsychotics in the prevention of hospitalization after lithium discontinuation in bipolar disorder. Eur. Neuropsychopharmacol. 61, 36–42 (2022).
-
Pacchiarotti, I. et al. Long-acting injectable antipsychotics (LAIs) for maintenance treatment of bipolar and schizoaffective disorders: a systematic review. Eur. Neuropsychopharmacol. 29, 457–470 (2019).
-
Prajapati, A. R., Wilson, J., Song, F. & Maidment, I. Second-generation antipsychotic long-acting injections in bipolar disorder: systematic review and meta-analysis. Bipolar Disord. 20, 687–696 (2018).
-
Keramatian, K., Chakrabarty, T. & Yatham, L. N. Long-acting injectable second-generation/atypical antipsychotics for the management of bipolar disorder: a systematic review. CNS Drugs 33, 431–456 (2019).
-
Taipale, H. & Tiihonen, J. Registry-based studies: what they can tell us, and what they cannot. Eur. Neuropsychopharmacol. 45, 35–37 (2021).
-
Kim, A. M., Salstein, L. & Goldberg, J. F. A systematic review of complex polypharmacy in bipolar disorder: prevalence, clinical features, adherence, and preliminary recommendations for practitioners. J. Clin. Psychiatry 82, 20r13263 (2021).
-
Grunze, H. et al. The World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the biological treatment of bipolar disorders: update 2012 on the long-term treatment of bipolar disorder. World J. Biol. Psychiatry 14, 154–219 (2013).
-
Hui Poon, S., Sim, K. & Baldessarini, R. J. Pharmacological approaches for treatment-resistant bipolar disorder. Curr. Neuropharmacol. 13, 592–604 (2015).
-
Kimmel, S. E., Calabrese, J. R., Woyshville, M. J. & Meltzer, H. Y. Clozapine in treatment-refractory mood disorders. J. Clin. Psychiatry 55, 91–93 (1994).
-
Calabrese, J. R. et al. Clozapine for treatment-refractory mania. Am. J. Psychiatry 153, 759–764 (1996).
-
Ciapparelli, A. et al. Clozapine for treatment-refractory schizophrenia, schizoaffective disorder, and psychotic bipolar disorder: a 24-month naturalistic study. J. Clin. Psychiatry 61, 329–334 (2000).
-
Green, A. I. et al. Clozapine in the treatment of refractory psychotic mania. Am. J. Psychiatry 157, 982–986 (2000).
-
Zarate, C. A. J., Tohen, M., Banov, M. D., Weiss, M. K. & Cole, J. O. Is clozapine a mood stabilizer?. J. Clin. Psychiatry 56, 108–112 (1995).
-
Suppes, T. et al. Clinical outcome in a randomized 1-year trial of clozapine versus treatment as usual for patients with treatment-resistant illness and a history of mania. Am. J. Psychiatry 156, 1164–1169 (1999).
-
Benedetti, A. et al. Augmentation of clozapine with aripiprazole in severe psychotic bipolar and schizoaffective disorders: a pilot study. Clin. Pract. Epidemiol. Ment. Health 6, 30–35 (2010).
-
Tiihonen, J. et al. Association of antipsychotic polypharmacy vs monotherapy with psychiatric rehospitalization among adults with schizophrenia. JAMA Psychiatry 76, 499–507 (2019).
-
Macfadden, W. et al. A randomized, double-blind, placebo-controlled study of maintenance treatment with adjunctive risperidone long-acting therapy in patients with bipolar I disorder who relapse frequently. Bipolar Disord. 11, 827–839 (2009).
-
Quiroz, J. A. et al. Risperidone long-acting injectable monotherapy in the maintenance treatment of bipolar I disorder. Biol. Psychiatry 68, 156–162 (2010).
-
Tanskanen, A. et al. From prescription drug purchases to drug use periods—a second generation method (PRE2DUP). BMC Med. Inform. Decis. Mak. 15, 21 (2015).
-
Allison, P. D. Fixed Effects Regression Models (SAGE, 2009).
-
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B 57, 289–300 (1995).
-
Prada-Ramallal, G., Takkouche, B. & Figueiras, A. Bias in pharmacoepidemiologic studies using secondary health care databases: a scoping review. BMC Med. Res. Methodol. 19, 53 (2019).
Acknowledgements
This study was funded by the Swedish Research Council for Health, Working Life and Welfare, FORTE (2021–01079). The Finnish data were supported by the Finnish Ministry of Social Affairs and Health through the developmental fund for Niuvanniemi Hospital. J.L. was funded by the Finnish Medical Association (grant 7709). H.T. was funded by the Sigrid Jusélius Foundation. We utilized data from the REWHARD consortium supported by the Swedish Research Council (grant number 2021–00154). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Funding
Open access funding provided by Karolinska Institute.
Ethics declarations
Competing interests
J.T., H.T., E.M.-R. and A.T. participated in research projects funded by grants from Janssen to their employing institution. J.T. has been a consultant and/or adviser to and/or has received honoraria from Eli Lilly, Evidera, HLS Therapeutics, Janssen-Cilag, Lundbeck, Mediuutiset, Orion, Otsuka, Sidera, Sunovion and WebMed Global. H.T. reports personal fees from Gedeon Richter, Janssen, Lundbeck and Otsuka. M.L. is a board member of Genomi Solutions Ltd and Springflux Ltd and has received honoraria from Sunovion, Orion Pharma, Camurus, Lundbeck, Otsuka Pharma, Recordati, Janssen and Janssen-Cilag and research funding from the Finnish Cultural Foundation and the Emil Aaltonen Foundation. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lieslehto, J., Tiihonen, J., Ármannsdóttir, B. et al. Comparative effectiveness of treatment strategies for bipolar disorder during and after lithium treatment. Nat. Mental Health (2026). https://doi.org/10.1038/s44220-026-00645-1
-
Received:
-
Accepted:
-
Published:
-
Version of record:
-
DOI: https://doi.org/10.1038/s44220-026-00645-1



Leave a Reply